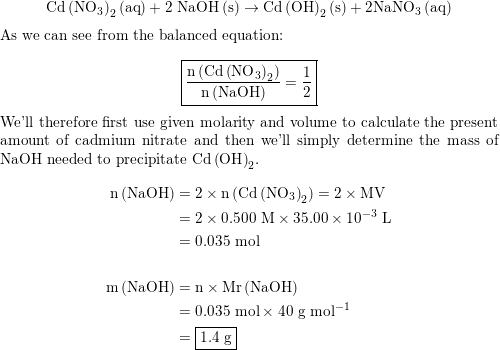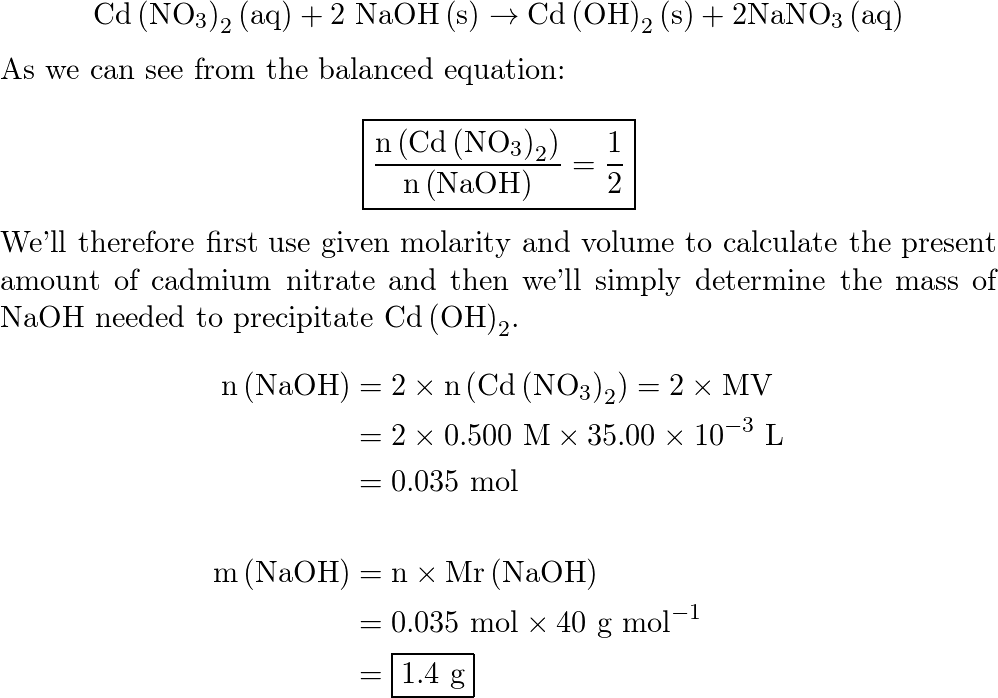Ni 2+ and Cd 2+ biosorption (mg•g −1 ) by NaOH-treated and untreated... | Download Scientific Diagram

Insights into the removal of Cd and Pb from aqueous solutions by NaOH–EtOH-modified biochar - ScienceDirect
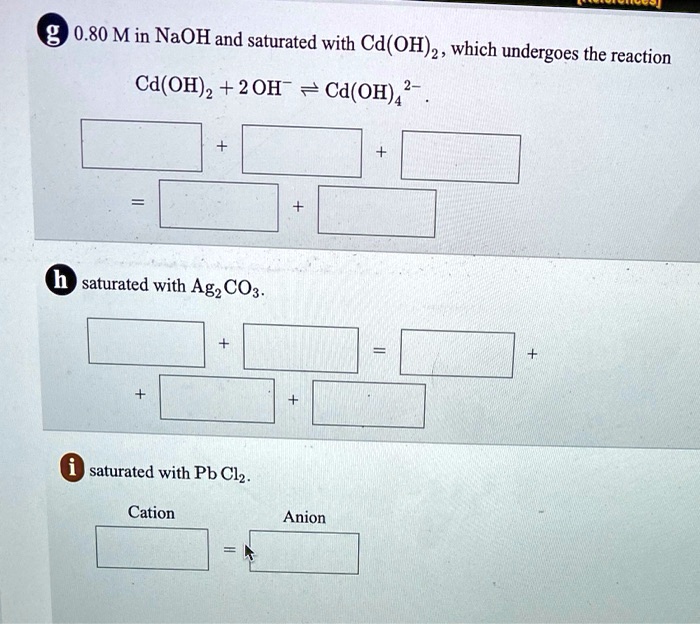
SOLVED: 0.80 M in NaOH and saturated with Cd(OH) 2, which undergoes the reaction Cd(OH)2 + 2 OH 7 Cd(OH)4> saturated with Ag2CO3. saturated with Pb Clz Cation Anion

Relationship between changes of extractability of Cd with NaOH and DTPA... | Download Scientific Diagram

Given the cell: `Cd(s)|Cd(OH)_2(s)|NaOH(aq,0.01M)|H_2(g,1bar)|Pt(s)` with `E_(cell)=0.0V.ifE_(Cd... - YouTube

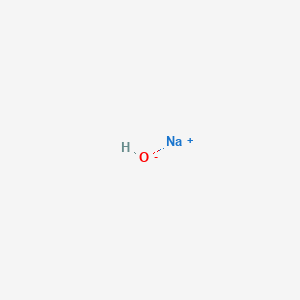
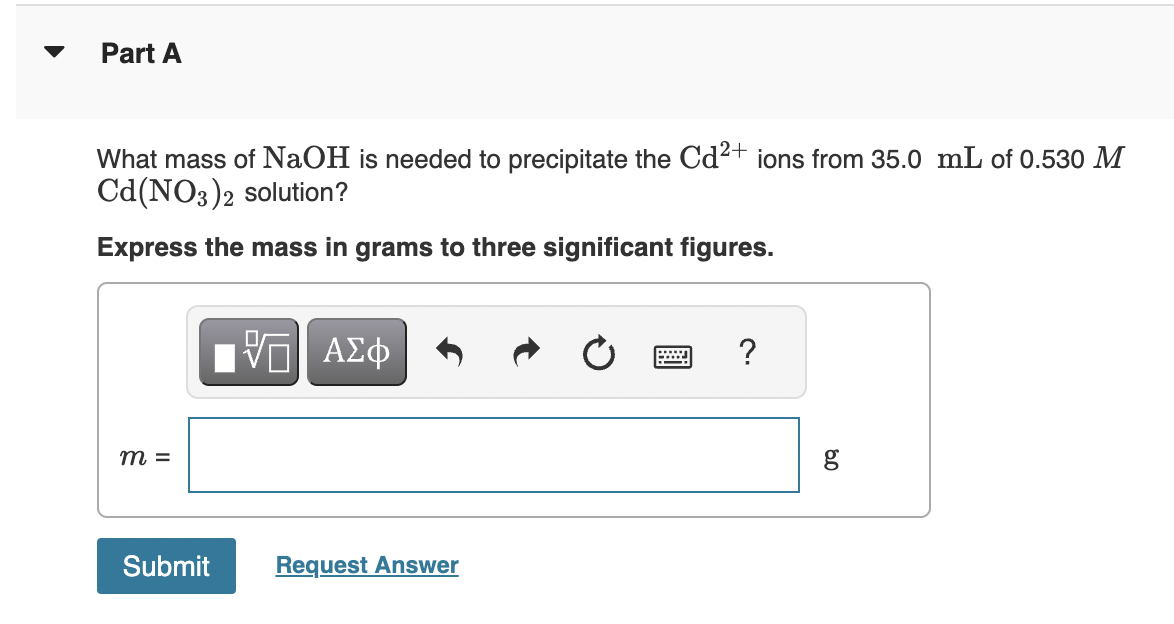
SOLVED: What mass of NaOH is needed to precipitate the Cd2+ ions from 32.0 mL of 0.550 M Cd(NO3)2 solution?

OneClass: 1. Let us assume that Cd(OH)2(s) is completely insoluble, which signifies that the precipit...

Given the cell: Cd(s)|Cd(OH)2(s)|NaOH(aq, 0.01 M)|H2(g, 1 bar)|Pt(s) with Ecell = 0.0 V . If E^oCd^2 + |Cd = - 0.39 V , then Ksp of Cd(OH)2 is:

Reduction of CD as a function of basic pH (Ca(OH)2 and NaOH) with a... | Download Scientific Diagram

Given the cell: Cd(s)|Cd(OH)2(s)|NaOH(aq, 0.01 M)|H2(g, 1 bar)|Pt(s) with Ecell = 0.0 V . If E^oCd^2 + |Cd = - 0.39 V , then Ksp of Cd(OH)2 is:

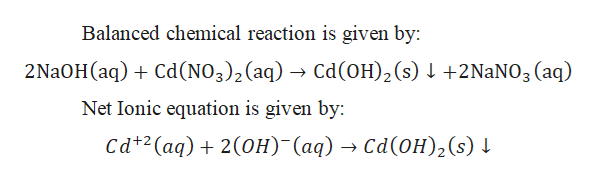
![ANSWERED] If 4 moles of NaOH react with 1 mole of C... - Physical Chemistry ANSWERED] If 4 moles of NaOH react with 1 mole of C... - Physical Chemistry](https://media.kunduz.com/media/sug-question/raw/47412038-1658679986.4827924.jpeg)